
I’m exhausted today still recovering from the aftermath of chemo’s carpet bombing but I wanted to tell you all about what is going to happen tomorrow and the best description I’ve come across is from the prestigious National Institute of Health’s website so please forgive me for plagiarising their words here (but at least you know it’s well sourced!). More, for those who want it, can be found at www.cancer.gov
For years, the foundations of cancer treatment were surgery, chemotherapy, and radiation therapy. Over the past two decades, targeted novel and often oral therapies, drugs that target cancer cells by homing in on specific molecular changes seen primarily in those cells—have also cemented themselves as standard treatments for many cancers. That was what led to venetoclax (targeting BCL2) and ibrutinib (targeting BTK) – the two drugs which have helped me to reach this point.
But over the past several years, immunotherapy—therapies which enlist and strengthen the power of a patient’s immune system to attack tumors—has emerged as what many in the cancer community now call the “fifth pillar” of cancer treatment. I’ve also had two of these treatments already both rituximab and obinutuzimab – two monoclonal antibodies which, as I describe to my children, put tails on the dodgy cancer cells and force my asleep-on-the job immune system to finally kick into gear to kill them.
However a new rapidly emerging immunotherapy approach is called adoptive cell transfer (ACT): collecting and using patients’ own immune cells to treat their cancer. There are several types of ACT but, thus far, the one that has advanced the furthest in clinical development is called CAR T-cell therapy.
Until recently, the use of CAR T-cell therapy was restricted to small clinical trials, largely in patients with advanced blood cancers. But these treatments have nevertheless captured the attention of researchers and the public alike because of the remarkable responses they have produced in some patients—both children and adults—for whom all other treatments had stopped working. If you want to see something that will really make you cry watch this video featuring Emily Whitehead – the first child ALL patient ever to have CAR-T and featuring the doctors who transformed its use
In 2017, two CAR T-cell therapies were approved by the Food and Drug Administration (FDA), one for the treatment of children with acute lymphoblastic leukemia (ALL) and the other for adults with advanced lymphomas (DLBCL). Nevertheless, researchers caution that, in many respects, it’s still early days for CAR T cells and other forms of ACT, including questions about whether they will ever be effective against solid tumors like breast and colorectal cancer.
And in January this year one of those CAR-Ts was approved here for lymphoma patients
https://www.petermac.org/car-t
I just happened to be at the Peter Mac for the press conference for work and ended up featuring in the story on behalf of blood cancer patients – little did I know I would be having this treatment in a trial only 6 months later.
CAR T cells are the equivalent of “giving patients a living drug,” explains Renier J. Brentjens, M.D., Ph.D., of Memorial Sloan Kettering Cancer Center in New York, an early leader in the CAR T-cell field. As its name implies, the backbone of CAR T-cell therapy is T cells, which are often called the workhorses of the immune system because of their critical role in orchestrating the immune response and killing cells infected by pathogens.
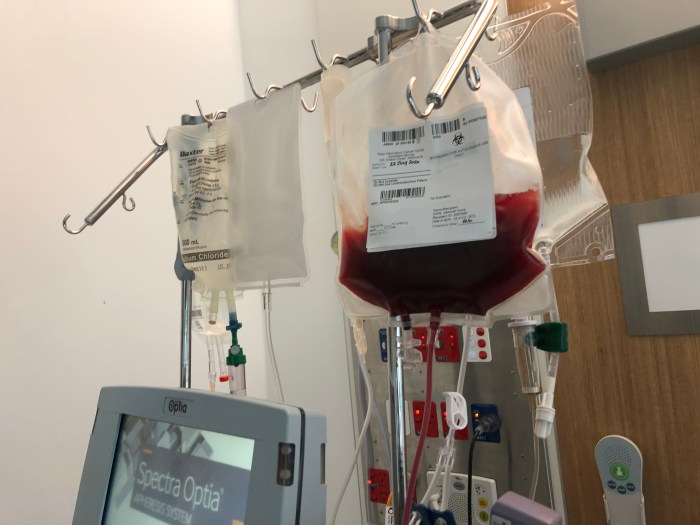
The therapy requires drawing blood from patients and separating out the T cells which is why on a cold July day I was in the Peter Mac having my T-cells harvested in the apheresis unit after extensive screening for a brand new clinical trial of CAR-T for CLL, the only one in the world that I would currently be eligible for and one that was at my local hospital rather than on the other side of the planet with 10 places available for patients with a poor prognosis who have been on ibrutinib for 6 months with stable disease. As far as I’m concerned if ever there was a sign that I should be doing something it was this.
The harvesting itself was an incredible procedure where a large (don’t look) needle was put in a vein in one arm, the blood siphoned through a machine which looked like a washing machine and then returned to me minus the T-cells they needed. The Senior haematology nurse who performed this procedure was constantly adjusting the machine to get the right number of cells – it was fascinating. The whole thing took 3 hours, was uncomfortable and a little unpleasant but a rather small price to pay for what comes next.
I received a phone call from James in the lab that night telling me that they needed 1 million of one type of cell and got 1.95 million and 2 million of another type and got 3.26 million (I was still a bit groggy but that was as much as I understood!). Funny that of all these photos taken on the day my kids liked the esky best – they liked the Hazard label! So that was back in July and, in the middle of covid lockdown, my cells winged their way to the US to be genetically engineered using a disarmed virus to produce receptors on their surface called chimerical antigen receptors, or CARs. These special receptors are synthetic molecules, they don’t exist naturally, but they allow the T cells to recognize and attach to a specific protein, or antigen, on tumor cells. And in my case they will target an antigen found on B cells called CD19 . Once my collected T cells finished being engineered to express the antigen-specific CAR, they were “expanded” in the laboratory into the hundreds of millions and that is what took place on my manufacture date which was August the 14th in a lab thousands of miles away.
A vial of precious cells arrived back here last week (an approved version of CAR-T in the US costs upwards of $500k so I am incredibly grateful for this trial and the fact, in Australia there is no cost involved for me). The final step happens tomorrow – the infusion of the CAR T cells into me (after a “lymphodepleting” chemotherapy regimen has been completed – boy did I tick that one – I’d forgotten how grim chemo is!). If all goes as planned, the engineered cells will further multiply in my body and, with guidance from their engineered receptor, recognize and kill cancer cells that harbor the antigen on their surface – and if they get the very last cell my incurable cancer will once and for all be cured! And that’s what I’m focusing on tonight ahead of my infusion tomorrow.
There is substantial risk with this procedure – I’ve lost a good friend within hours of her infusion a couple of years ago, there are neurotoxic side effects and it’s not going to be a walk in the park but I don’t want to dwell. What I will do though is share a couple of interviews I’ve done on the subject. One with my good friend Dr Brian Koffman whom I interviewed in Stockholm only 10 weeks after he had his CAR-T procedure and made it to the European Haematology Association conference there. And one with leading CAR-T researcher Dr Tanya Siddiqi who is running a trial for CAR-T in CLL at the City of Hope hospital in California – it was the interviews with her over a couple of years which have driven me most to try to get this treatment at a stage in my disease when I am most likely to survive it and benefit from it but more on that in my next update.





Dearest Deborah, what a journey and what an incredible lady you are. I wish you all the good luck in the world for your pioneering treatment. I’m sure you are in the best medical hands for this. Hang on in there, my friend. Sending you so much love. Sam xxxx
LikeLike
Thanks gorgeous – exciting and terrifying – a bit like being on-air! Hope you’re well – one of these days we’ll catch up x
LikeLike
Nice overview of the history of CAR-T. Plus all your personal details. Surprised you are so coherent. You will get through this. Brian http://cllsociety.org
LikeLike
Thanks so much Brian – Day 0 eek! This seems to have come around so quickly – thanks for the comforting words and for being with me on so much of this journey – looking forward to a hug next time we meet (in a world where there is a Covid vaccine and we don’t have to worry about that anymore either!).
LikeLike
Take care! Thinking if you! ❤️
LikeLike
Thanks Carol x
LikeLike
All the very best! You are a strong, determined person. I am sending you all my best wishes. I will follow your blog with keen interest.
LikeLike
Sending love and please know you have friends surrounding you. You will get through this.
LikeLike
Deborah, wishing you the very best today and in the days ahead. I hope it will reassure you to know that my husband underwent successful CAR T-cell therapy for his CLL in January at Peter Mac, now coming up for his nine-month review.
LikeLike
Oh that’s wonderful thanks for letting me know Donette – the nurses are slightly intrigued by us – they only have a few of us each month – it is exciting – I’m so pleased it’s worked well for your husband – these are the stories we need to hear!
LikeLike
Deb my love , you’re a fighter! I know you can get through this. Look how far you’ve come already!! I’m loving your updates helping us understand. Sending all my love and positive thoughts to you. Xxxx
LikeLike
Thanks Harriet – can’t wait to celebrate with you over Summer x
LikeLike
Good luck and all our thoughts and prayers are with you.
LikeLike
Thanks Elizabeth x
LikeLike
Hi Deborah, I came across your blog via Health Unlocked. As a caregiver of a patient with Mantel Cell Lymphoma, I’m so glad I’ve discovered you! My partner will be having CAR-T therapy on 6th October. He is scared and very nervous, as I’m sure you are going into this. I have just shown him your video with Dr. Brian Koffman, which is just brilliant and has answered a lot of questions. It has helped my partner to see and listen to others who have gone through the treatment and come out the other end. I will be following your treatment via your blog very closely. Thank you so much for doing it, as I’m sure there will be times it will be difficult. Your positivity and drive will be a tremendous help for yourself and others. I and partner appreciate everything you have done and contiue to do. Best wishes.
LikeLike
Oh this is lovely thanks Pauline. My nurse just asked what I was so busy with and I showed her – I’m not sure I know how to switch off and I think it’s important to record this for others as much as possible. I just looked at Brian’s blog and I received my CAR-T cells exactly 30 months to the day that he received his – he has helped me enormously in my decision and it wasn’t an easy one to make but I hope your husband finds, like me, that once the cells are in a lot of the anxiety dissipates – I think it must be that there’s not much I can do anyway (other than walk around the ward to stop them giving me another of those anti clotting jabs in my stomach – seriously of everything they’ve done to me this week that was by far the most painful!). It’s still early days but I’m now 32 hours post infusion and haven’t had a single reaction – I’m on a dose escalation study of a new form of CAR-T which is younger when it’s infused but there’s lots of data with the existing CAR-Ts. I do hope this is your husband and my cures – I’m sure, like me, he has been through the mill and it’s time for a curative option. Stay in touch and let him know he can reach out to me at any time. Thanks for taking the time to post x
LikeLike
One thing I would say about Brian’s experience and those of people from that era is that they now know how to treat CRS and neurotoxicity more than they did then so, while I’m still prepared for a bit of a torrid time at some stage, I’m optimistic it won’t be as bad as what he and others suffered then – thank goodness for great caregivers and you are obviously one x
LikeLike